Abstract
Background: Cytoreduction with hydroxyurea (HU) is the recommended first-line (1L) treatment for high-risk polycythemia vera (PV) (age ≥60 years or history of thrombosis), but many patients (pts) have suboptimal response to HU. This study describes characteristics, blood counts, treatments, and clinical outcomes among pts with PV, including those with suboptimal response to HU, in 2 large networks of US community oncology practices.
Methods: This was a retrospective observational study of adult pts with a documented new diagnosis of PV in the iKnowMed electronic health record database (01Jan2008-31Jan2020), which is implemented in The US Oncology Network and non-Network practices. Pts with ≥2 postdiagnosis visits during the study observation period (01Jan2008-31Jan2021) were included in the analysis. Suboptimal response to HU was defined as meeting ≥1 of the following criteria after ≥3 months of treatment: (1) white blood cell (WBC) count >10 × 109/L, (2) platelet count >400 × 109/L, and/or (3) hematocrit (HCT) >45%. Data on demographic/clinical characteristics and prior treatment were summarized with descriptive statistics. Duration of therapy and overall survival were assessed via the Kaplan-Meier method, which censored pts without subsequent treatment discontinuation/death records at the end of the study observation period.
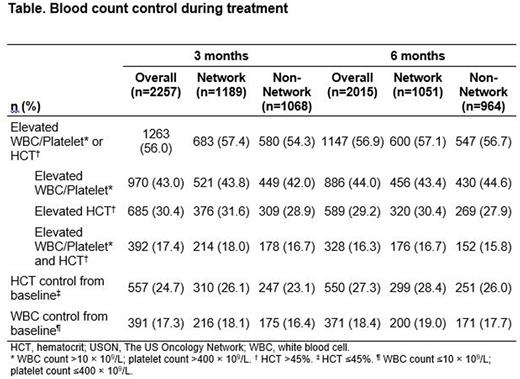
Results: 5871 pts (The US Oncology Network, n=3022; non-Network, n=2849) met all eligibility criteria and were included in the analysis. Mean (SD) age at diagnosis was 66.1 (13.6) years (70% ≥60 years), 44% of pts were female, 87% were White, and 49% were current or former tobacco users. At diagnosis, 6% of pts had a history of thrombotic event, and 71% had high-risk PV; 38% (1535/4021), 34% (1361/4014), and 67% (2696/4010) of evaluable pts had elevated WBC (>10 × 109/L), platelets (>400 × 109/L), and HCT (>45%), respectively. Over half of pts with high-risk PV (55%; 2303/4185) received a cytoreductive agent as 1L treatment (90% HU), and 45% did not receive cytoreductive therapy. Among those with low-risk PV, 32% (540/1686) received a cytoreductive agent as 1L treatment (89% HU), and 68% did not receive cytoreductive therapy. Five-year survival probability was 82% among all pts, 79% for pts with high-risk PV (age ≥60 years or history of thrombosis), and 96% for those with low-risk PV. Overall, 56% of patients had a suboptimal response for blood count control after 3 months of treatment; this remained roughly the same at 6 months (Table).
Among pts who received 1L HU, median (Q1, Q3) time from diagnosis to HU initiation was 29.0 (0.0, 252.5) days for those with high-risk PV and 103.0 (0.0, 683.5) days for those with low-risk PV; median (Q1, Q3) duration of HU was 26.7 (8.1, 54.6) and 35.6 (9.3, 68.5) months, respectively. Among pts treated with 1L HU who had evaluable blood counts, 42% (1078/2548) had a suboptimal response at 3 months, 64% of whom (691/1078) continued to have a suboptimal response at 6 months (WBC >10 × 109/L and/or platelets >400 × 109/L, 50%; HCT >45%, 36%). Median duration of 1L HU treatment for pts with suboptimal response was 26.6 (95% CI: 24.0, 29.3) months. Among pts with suboptimal response to 1L HU, 5-year survival probability was 82% overall (high risk, 78%; low risk, 99%). Pts with suboptimal response appeared to have a lower 5-year survival probability than pts with optimal response (81.5% [95% CI: 77.7%, 85.5%] vs 84.3% [95% CI: 81.2%, 87.4%]). This difference between pts with suboptimal vs optimal response was statistically significant in non-Network (74.5% [95% CI: 67.7%, 82.0%] vs 85.1% [95% CI: 81.1%, 89.3%]; P=0.008) but not in The US Oncology Network practices (86.7% [95% CI: 82.3%, 91.2%] vs 83.6% [95% CI: 79.1%, 88.3%]; P=0.109). Besides this difference observed in the 5-year survival probability, pt characteristics and outcomes were similar between the 2 networks, although additional data (eg, pt/disease characteristics; phlebotomy use, supportive care) are needed to better understand differences between networks.
Conclusions: The prevalence of elevated blood counts at diagnosis and follow-up while on HU treatment is consistent with previous real-world evidence. About 1 in 6 pts with a suboptimal response to HU died within 5 years, and there was a trend toward shorter survival among those with suboptimal response compared with pts with blood count control. HU dose adjustments or alternative treatments may be needed for pts with suboptimal response to HU.
Disclosures
Lyons:Incyte Corporation: Consultancy, Other: Research Consulting Services; Texas Oncology/The US Oncology Network: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company, Other: Leadership; Celgene: Honoraria. Aguilar:Ontada: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company; Incyte: Consultancy, Other: Research Consulting Services to Incyte. Sudharshan:Ontada: Current Employment; Incyte: Consultancy, Other: Research Consulting Services to Incyte. Venkatasetty:Ontada: Current Employment; Incyte: Consultancy, Other: Research Consulting Services to Incyte. Ndukum:Ontada: Current Employment; Incyte: Consultancy, Other: Research Consulting Services to Incyte. Zackon:Incyte: Consultancy, Other: Research Consulting Services to Incyte; Ontada: Current Employment; Epizyme: Consultancy; Morphosys: Consultancy. Yu:Incyte Corporation: Current Employment, Current equity holder in publicly-traded company.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal